- Home
- About
- Products
- Contact Temperature Sensors
- Cables & Wires
- Mineral Insulated Cables
- Nickel & Thermocouple Alloy
- Industrial Heaters
- Heating Cables and Mats
- Non Contact Temperature Sensors
- Industrial and R&D Furnaces
- Temperature Calibrators
-
Circulating Chiller
- Services
-
Special Products
- Thermal Profiling System
- Industries
- Resources
- Contact Us
- Shop
Temperature and Temperature Scales
The temperature of a substance is the degree of hotness or coldness of the substance. A hot substance is said to have a high temperature whereas a cold substance is said to have a low temperature. Therefore, the temperature of a substance is an indication of the average kinetic energy of the molecules of the substance. Heat always flow from a body at higher temperature to the body at lower temperature. So, we can also say that temperature of a body is the property which governs the flow of heat.
It can easily be demonstrated as follows: When two objects of the same material are placed together, the object with higher temperature cools while the cooler object gets warmer until a point is reached after which no more changes occurs.
Thermal Equilibrium
Heat energy flows from a body at higher temperature to another body at lower temperature. In other words, heat flows from a hotter to the colder body. The heat energy keeps flowing from the hotter to the cooler body until the temperature of both the bodies become equal. At this stage, the two bodies are said to be in thermal equilibrium. When two bodies attain the same temperature, they are said to be in thermal equilibrium with each other because then no heat flows from one body to another. The temperature thus reached is called as equilibrium temperature.
Therefore, now we can say that Temperature is that quantity which is same for both the system when they are in thermal equilibrium.
Zeroth Law Of Thermodynamics
ZEROTH law of thermodynamics state that "If two systems are separately in thermal equilibrium with a third, then they must also be in thermal equilibrium with each other, and they all have the same temperature regardless of the kind of the system they are."
This law can be restated as follows:
If there are three or more than three systems which when taken together are in thermal contact and in thermal equilibrium as well, then any of the two system taken together are in equilibrium with one another.
Temperature Scale
One of the first attempts to make a standard temperature scale occurred in very past, when Galen, in his medical writings, proposed a standard "neutral" temperature made up of equal quantities of boiling water and ice; on either side of this temperature were four degrees of heat and four degrees of cold, respectively. The earliest devices used to measure the temperature were called thermoscopes. They consisted of a glass bulb having a long tube extending downward into a container of coloured water, although Galileo in 1610 is supposed to have used wine. Some of the air in the bulb was expelled before placing it in the liquid, causing the liquid to rise into the tube. As the remaining air in the bulb was heated or cooled, the level of the liquid in the tube would vary reflecting the change in the air temperature. An engraved scale on the tube allowed for a quantitative measure of the fluctuations. The air in the bulb is referred to as the thermometric medium, i.e. the medium whose property changes with temperature.
In 1641, the first sealed thermometer that used liquid rather than air as the thermometric medium was developed for Ferdinand II, Grand Duke of Tuscany. His thermometer used a sealed alcohol-in-glass device, with 50 "degree" marks on its stem but no "fixed point" was used to zero the scale. These were referred to as "spirit" thermometers.
Robert Hook, Curator of the Royal Society, in 1664 used a red dye in the alcohol. His scale, for which every degree represented an equal increment of volume equivalent to about 1/500 part of the volume of the thermometer liquid, needed only one fixed point. He selected the freezing point of water. By scaling it in this way, Hook showed that a standard scale could be established for thermometers of a variety of sizes. Hook's original thermometer became known as the standard of Gresham College and was used by the Royal Society until 1709.
(The first intelligible meteorological records used this scale). In 1702, the astronomer Ole Roemer of Copenhagen based his scale upon two fixed points: snow (or crushed ice) and the boiling point of water, and he recorded the daily temperatures at Copenhagen in 1708- 1709 with this thermometer.
It was in 1724 that Gabriel Fahrenheit, an instrument maker of Däanzig and Amsterdam, used mercury as the thermometric liquid. Mercury's thermal expansion is large and fairly uniform, it does not stick to the glass, and it remains a liquid over a wide range of temperatures. Its silvery appearance makes it easy to read.
Fahrenheit described how he calibrated the scale of his mercury thermometer:
"Placing the thermometer in a mixture of salt ammoniac or sea salt, ice, and water a point on the scale will be found which is denoted as zero. A second point is obtained if the same mixture is used without salt. Denote this position as 30. A third point, designated as 96, is obtained if the thermometer is placed in the mouth so as to acquire the heat of a healthy man."
On this scale, Fahrenheit measured the boiling point of water to be 212. Later he adjusted the freezing point of water to 32 so that the interval between the boiling and freezing points of water could be represented by the more rational number 180.
Temperatures measured on this scale are designated as degrees Fahrenheit (°F). In 1745, Carolus Linnaeus of Upsula, Sweden, described a scale in which the freezing point of water was zero, and the boiling point 100, making it a centigrade (one hundred steps) scale each step was called "degree". Anders Celsius (1701-1744) used the reverse scale in which 100 represented the freezing point and zero the boiling point of water, still, of course, with 100 degrees between the two fixed points.
In 1948 use of the Centigrade scale was dropped in favor of a new scale using degrees Celsius (° C). The Celsius scale is defined by the following two items that will be discussed later in this essay:
- The triple point of water is defined to be 0.01°
- A degree Celsius equals the same temperature change as a degree on the ideal-gas
On the Celsius scale the boiling point of water at standard atmospheric pressure is 99.975 C in contrast to the 100 degrees defined by the Centigrade scale.
To convert from Celsius to Fahrenheit: multiply by 1.8 and add 32. ° F = 1.8° C + 32 ° K = ° C + 273.
In 1780, J. A. C. Charles, a French physician, showed that for the same increase in temperature, all gases exhibited the same increase in volume. Because the expansion coefficient of gases is nearly the same, it is possible to establish a temperature scale based on a single fixed point rather than the two fixed- point scales, such as the Fahrenheit and Celsius scales. This brings us back to a thermometer that uses a gas as the thermometric medium.
In a constant volume gas thermometer a large bulb B of gas, hydrogen for example, under a set pressure connects with a mercury-filled "manometer" by means of a tube of very small volume. (The Bulb B is the temperature-sensing portion and should contain almost all of the hydrogen). Raising or lowering the mercury reservoir R may adjust the level of mercury at C. The pressure of the hydrogen gas, which is the "x" variable in the linear relation with temperature, is the difference between the levels D and C plus the pressure above D. P.Chappuis in 1887 conducted extensive studies of gas thermometers with constant pressure or with constant volume using hydrogen, nitrogen, and carbon dioxide as the thermometric medium. Based on his results, the Comité International des Poids et Measures adopted the constant-volume hydrogen scale based on fixed points at the ice point (0° C) and the steam point (100° C) as the practical scale for international meteorology. Experiments with gas thermometers have shown that there is very little difference in the temperature scale for different gases. Thus, it is possible to set up a temperature scale that is independent of the thermometric medium if it is a gas at low pressure. In this case, all gases behave like an "Ideal Gas" and have a very simple relation between their pressure, volume, and temperature: pV= (constant) T.This temperature is called the thermodynamic temperature and is now accepted as the fundamental measure of temperature. Note that there is a naturally defined zero on this scale - it is the point at which the pressure of an ideal gas is zero, making the temperature also zero. With this as one point on the scale, only one other fixed point need be defined.
In 1933, the International Committee of Weights and Measures adopted this fixed point as the triple point of water, the temperature at which water, ice, and water vapor coexist in equilibrium); its value is set as 273.16. The unit of temperature on this scale is called the Kelvin, after Lord Kelvin (William Thompson), 1824-1907, and its symbol is K (no degree symbol used).
To convert from Celsius to Kelvin, add 273. K = ° C + 273.
Thermodynamic temperature is the fundamental temperature; its unit is the Kelvin, which is defined as the fraction 1/273.16 of the thermodynamic temperature of the triple point of water.
In 1871, Sir William Siemens,proposed a thermometer whose thermometric medium is a metallic conductor whose resistance changes with temperature. The element platinum does not oxidize at high temperatures and has a relatively uniform change in resistance with temperature over a large range. The Platinum Resistance Thermometer is now widely used as a thermoelectric thermometer and covers the temperature range from about -260° C to 1235°C.
Relationship Between Different Temperature Scale
Sometimes it is required to change or convert the value of temperature from one temperature scale to another temperature scale, for this there is a requirement of relationship between different temperature scales. The relationship between different temperature scales is:
C-0/100 = F-32/180 = K-273/100 = Re-0/80 = Ra-491.67/180
Where, C, F, K, Re, Ra is denoting the different temperature scales. 0, 32, 273, 0, 491.67 is denoting the lower fixed points of different temperature scales & 100, 180, 100, 80, 180 is denoting the no. of divisions in different temperature scales.
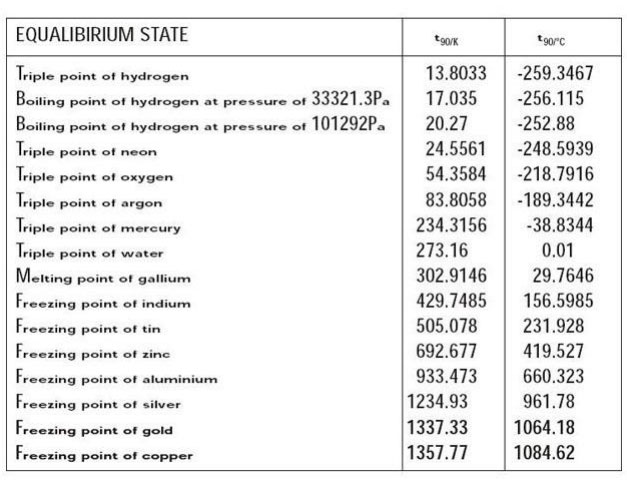
International Temperature Scale
The first internationally recognized temperature scale was the international temperature scale of 1927 ITS-27. Its purpose was to define procedures by which specified, high quality yet practical thermometry could be calibrated such that the values of temperature obtained from them would be concise and consistent instrument-to-instrument and sensor-to-sensor, while simultaneously approximating to the appropriate thermodynamic values within the limits of the technology available. This goal remains intact today.ITS-27 extended from just below the boiling point of oxygen, -200°C, to beyond the freezing point of gold, 1065°C. interpolation formulae were specified for platinum resistance thermometer calibrated at 0°C & at the boiling points of oxygen, water and sulphur (445°C). Above 660°C, the Pt-10% Rh vs. Pt thermocouple was specified for measurement. Above the gold point optical pyrometry was employed and the values of the fixed points were based on the best available gas thermometry data of the day.